UF Purification vs. Microfiltration, Which Process To Choose For Your Application?
Anyone who has studied at the very least high school level biology (and paid attention) is familiar with the concept of a membrane, in particular a semi-permeable membrane. Biological living cells are wrapped in semi-permeable membranes that keep their functions separate from the surrounding environment. The semi-permeable aspect allows only certain ions and organic molecules to pass into or out of the cell. The membrane can be selective in either a passive or active capacity. The UF purification and microfiltration processes utilize a semi-permeable membrane to separate micro-contaminants from a water stream.
What is the difference between UF purification and microfiltration? We will first explain how a semi-permeable membranes works. Following this, we will explain the differences between the UF and microfiltration membrane treatment processes for water & wastewater treatment.
How does a semi-permeable membrane work?
One of the ways is through, active transportation across a membranes surface which occurs in a few different ways. Each one of these transports require the cell to expend a certain amount of energy to do so. One way is through transport channels that pull in and expel nutrients and metabolic wastes respectively. Another is endocytosis whereby the cell wall forms something like a pseudo mouth, wrapping around an external object and then budding off within the cell as a vesicle. Its opposite is exocytosis. Internal vesicles fuse with the membrane and its contents are secreted out into the surrounding solution.
Another way is through, passive mechanisms that are known as diffusion and osmosis. Diffusion is the movement of ions and molecules from areas of high concentration to areas of low concentration in order to create a state of equilibrium on both sides of the membrane. As these ions move about, they create an osmotic pressure difference. Osmosis works opposite diffusion, seeking to create equilibrium by moving a solvent fluid (typically water) to the higher concentration area.
The passive diffusion/osmosis process is a mechanism that is easy to replicate on a much larger scale. There are many potential applications for such technology, but in particular, is its usefulness in water & wastewater treatment. Micro and ultrafiltration purification are two such membrane technologies.They are very similar filtration/separation processes with a difference that makes each ideal for their own particular applications.
Microfiltration and UF purification are more alike than they are different. As mentioned in the introduction, they are both passive, membrane-based, separation technologies. These system processes work by applying differential pressure across a semi-permeable membrane and that pressure forces water and small particulate matter through the membrane pores while larger solids are retained on the other side.
These processes both also make for beneficial pre-treatment steps for reverse osmosis. Membranes need a lot of care, so they can last as long as possible without replacement. Filter pre-treatment reduces concentrations of larger solid particles and reduce chances of membrane fouling.
The membranes for these microfiltration and UF purification systems are also available in the same configurations. Plate and frame, tubular, hollow fiber, and spiral wound are possible options. These different configurations offer their own pros and cons. There are also different materials the membranes can be composed of, namely polymers and ceramic.
Similar Benefits:
No chemicals
Constant product quality regardless of feed quality
Compact
Similar costs:
Equipment
tanks, pumps, skids, controls, etc.
Construction materials
Water characterization
What is in the water/wastewater determines what needs to be done to treat it properly. More complex compositions or high concentrations of pollutants will require pre-treatment steps or more energy-intensive processes or more resilient membranes to handle these conditions. Low concentrations and simple contaminant compositions tend to require less pretreatment and therefore, reduce operational costs..
Flow rates
Higher flow rates are associated with higher capital & operational costs
Planning
Space requirements
Installation
Prepackaged vs Un-assembled systems
Shipping fees
Operational costs
The Differences
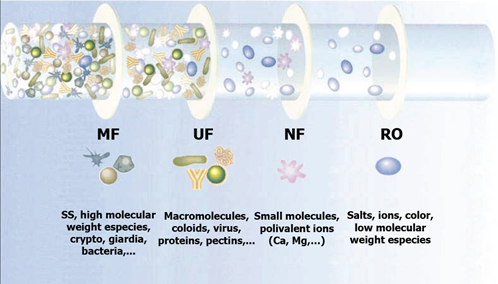
It all boils down to pore size. On the membrane separation scale, micro and ultrafiltration are coarser than nanofiltration and reverse osmosis, but are still finer than media filtration. Microfilter pores are within the range of 0.1 to 10 microns and ultrafiltration membrane pores within 0.01 to 0.1 microns. The method selected for a treatment system is based on the size of the smallest particles to be retained in the feedwater. The difference in their pore size determines the applications for which ultrafiltration purification or microfiltration treatment process would be the most applicable to be applied for the specific application.
Removal
Based on the pore size range of these two separation technologies, below is a list of some of the smallest pollutants that each technology is capable of removing or reducing from raw water streams.
Microfiltration (MF)
Algae
Bacteria
Pathogenic protozoa (Giardia lamblia and Crypotosporidium)
Sediment (sand, clay, certain complex metals/particles)
Ultrafiltration
All of the contaminants MF can remove plus:
Endotoxins
Plastics
Silica
Silt
Viruses
Applications
Both microfiltration and UF purification are useful for water/wastewater treatment in a broad range of industrial and commercial settings. This includes, the processing of many kinds of end products. Below, are only a few of the many possible applications for each membrane filter process.
Microfiltration
Cold sterilization of beverages and pharmaceuticals
Separating bacteria from water
Clarifying fruit juices, wine, or beer
Petroleum refining
Silt density index reduction for reverse osmosis
Virus removal from water
Separating oil/water emulsions
Removing pathogens from milk
Medical applications
Do you have any questions about micro or UF purification processes for your specific treatment application?
Contact the water treatment experts at Genesis Water Technologies, Inc. at 1-877-267-3699 or reach out to us by email at customersupport@genesiswatertech.com to discuss your treatment issues and the appropriate solution that will meet your specific needs.

